
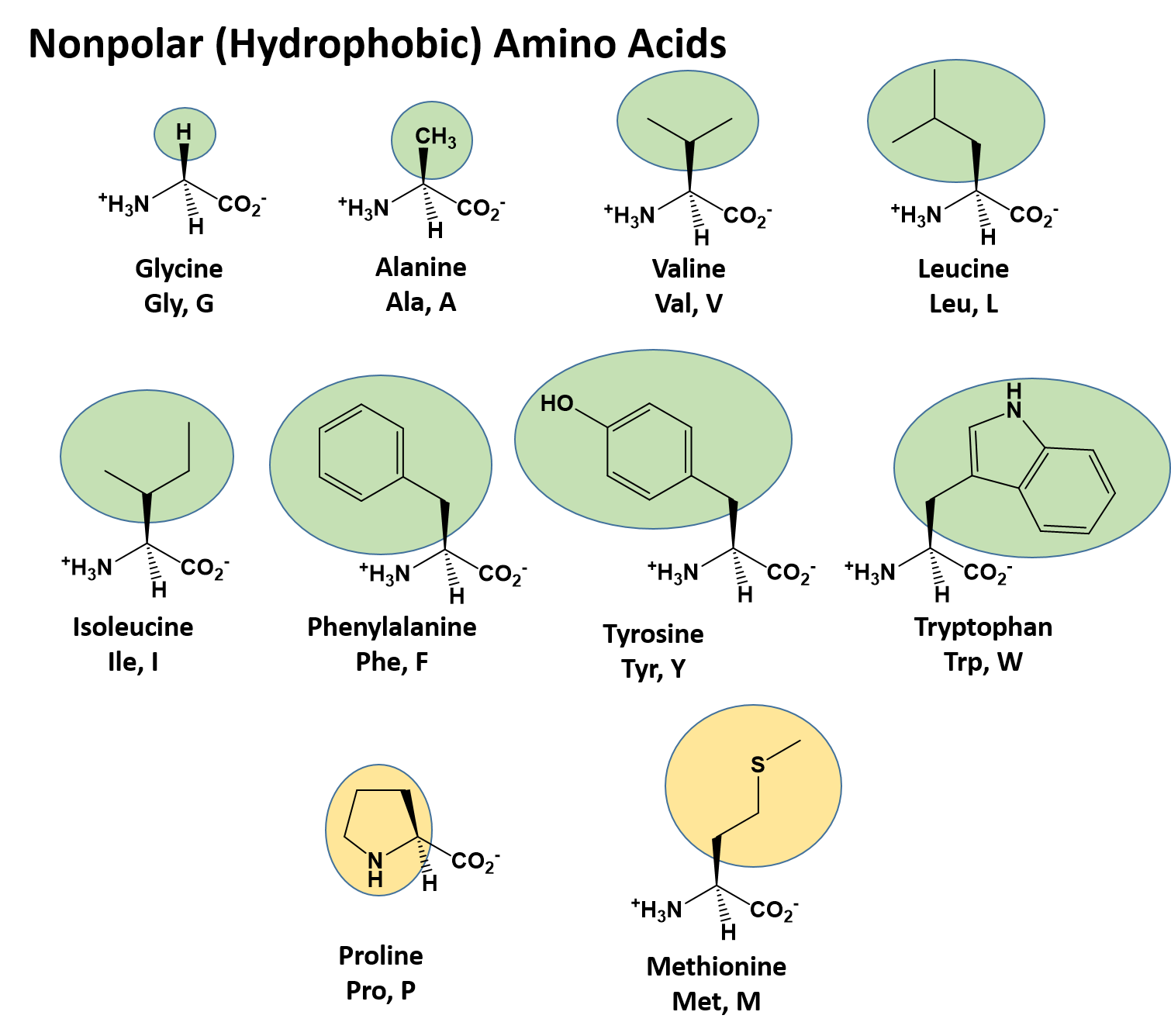
In the process, a molecule of water is released. The carboxyl group of one amino acid is linked to the amino group of the incoming amino acid. Peptide bond formation is a dehydration synthesis reaction. Essential amino acids refer to those necessary for construction of proteins in the body, although not produced by the body which amino acids are essential varies from organism to organism.įigure 3. They are known as essential amino acids, and in humans they include isoleucine, leucine, and cysteine. Just as some fatty acids are essential to a diet, some amino acids are necessary as well. For example, valine is known by the letter V or the three-letter symbol val. Proline is an exception to the standard structure of an animo acid since its amino group is not separate from the side chain (Figure 2).Īmino acids are represented by a single upper case letter or a three-letter abbreviation. Proline has an R group that is linked to the amino group, forming a ring-like structure. The side chains of lysine and arginine are positively charged, and therefore these amino acids are also known as basic amino acids. Amino acids such as valine, methionine, and alanine are nonpolar or hydrophobic in nature, while amino acids such as serine, threonine, and cysteine are polar and have hydrophilic side chains. For example, the amino acid glycine has a hydrogen atom as the R group. The chemical nature of the side chain determines the nature of the amino acid (that is, whether it is acidic, basic, polar, or nonpolar). Sometimes, positively and negatively charged amino acid side chains interact with one another in the interior of a protein, and polar or charged amino acid side chains that interact with a ligand can be found in the ligand binding pocket. In membrane proteins, nonpolar and hydrophobic amino acid side chains associate with the hydrophobic tails of phospholipids, while polar and charged amino acid side chains interact with the polar head groups or with the aqueous solution. Polar and charged amino acid residues (the remainder after peptide bond formation) are more likely to be found on the surface of soluble proteins where they can interact with water, and nonpolar (e.g., amino acid side chains) are more likely to be found in the interior where they are sequestered from water.
